Writing in the March 2026 edition of DIA’s Global Forum magazine, PharmCube’s Juan Valencia looks back on a record year for drug approvals in China, and what it means for the future of access and innovation in the world’s second largest market.
With the tremendous development of China’s innovative drug industry as backdrop, 2025 saw the number of new drugs approved by the National Medical Products Administration (NMPA)—the country’s regulatory authority—reach triple digits for the first time, an increase that was driven primarily by therapies developed domestically. The new entrants cover an array of different modalities, therapeutic areas, and developers, showcasing the maturity and increasing diversification of China’s pharmaceutical landscape.
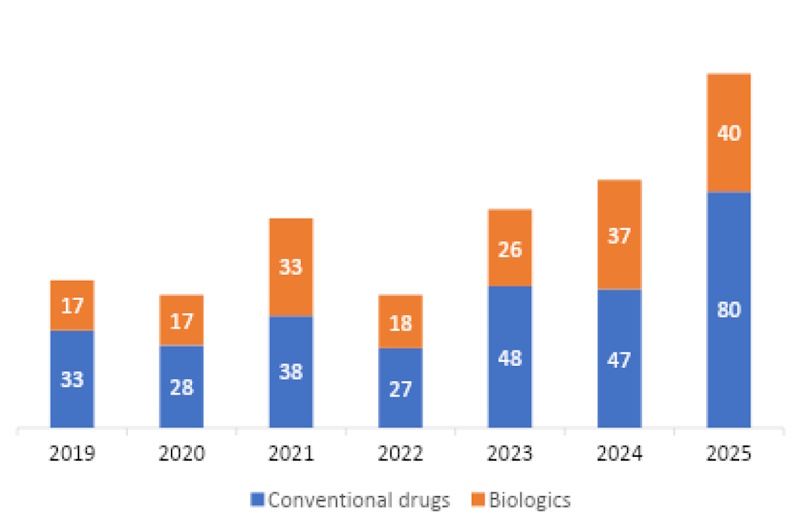
Number of new drugs approved by China’s NMPA between 2019 and 2025.
Source: PharmCube NextBiopharm® database.
Note: Throughout this article, “new drugs” refers to new molecular entities (NMEs) and their combinations, biological drugs, and vaccines that were approved for marketing in China for the first time, excluding biosimilars, new indications, and new dosage forms.



