Andrea Johnston,* Senior Phorum Director at BioPhorum,** argues that the biopharma industry’s rapid embrace of digital twins is being undermined by a fundamental problem: agreeing what a digital twin actually is. Drawing on fresh benchmarking data from leading manufacturers, Johnston contends that this lack of shared definition is slowing regulatory acceptance, limiting scalability, and constraining return on investment. She proposes a consensus framework and maturity model as a critical first step toward unlocking the technology’s full potential across the drug product lifecycle.
We cannot expect fragmented terminology to support a cohesive digital infrastructure
Leaders in pharmaceutical innovation have embraced the ‘digital twin’ concept with enthusiasm, recognising its profound potential to revolutionise everything from drug development to manufacturing operations.
Yet, despite this industry wide interest, we face a critical bottleneck, a lack of shared language and structured definition. This ambiguity is not merely an academic issue, it actively inhibits the widespread acceptance and adoption of digital twin technology, thereby stalling meaningful progress and preventing us from realising critical operational benefits.
The term itself has become a catch-all, sometimes confusingly used to encompass basic modelling or emulation. The issue is starkly illuminated by recent industry benchmarking. A BioPhorum survey of biomanufacturers such as GSK, Pfizer, Takeda, Merck and Co (MSD globally) and Sanofi, as well as research organisations and suppliers, reveals:
– Almost one third (27 percent) of respondents did not have a company definition of digital twins.
– Despite this acknowledged lack of definition, 48 percent of respondents stated that their company currently utilises digital twin technologies.
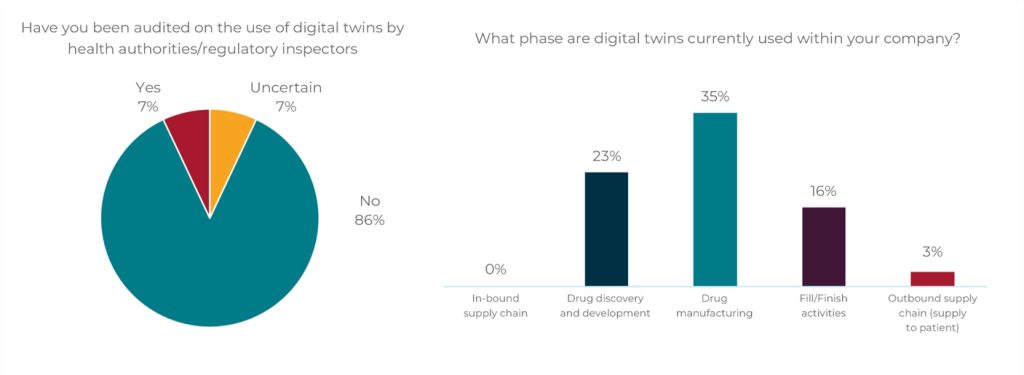
– Perhaps most telling, 86 percent of respondents said they had not been audited on the technology, implying it was not being used for critical regulatory processes.
We are in a peculiar position, with half the industry implementing a technology that nearly a third cannot formally define, and which is largely circumventing the scrutiny required for critical applications. This high amount of variability in understanding, terminology, and definition will invariably inhibit the widespread scaling and acceptance of the technology.
BioPhorum Benchmark Survey: Phase Deployment and Audit of Digital Twins
The key to unlocking progress is providing a single, universally accepted framework. The BioPhorum team has already engaged in this collaborative effort, proposing a consensus definition of a digital twin tied to the maturity levels of the technology itself.
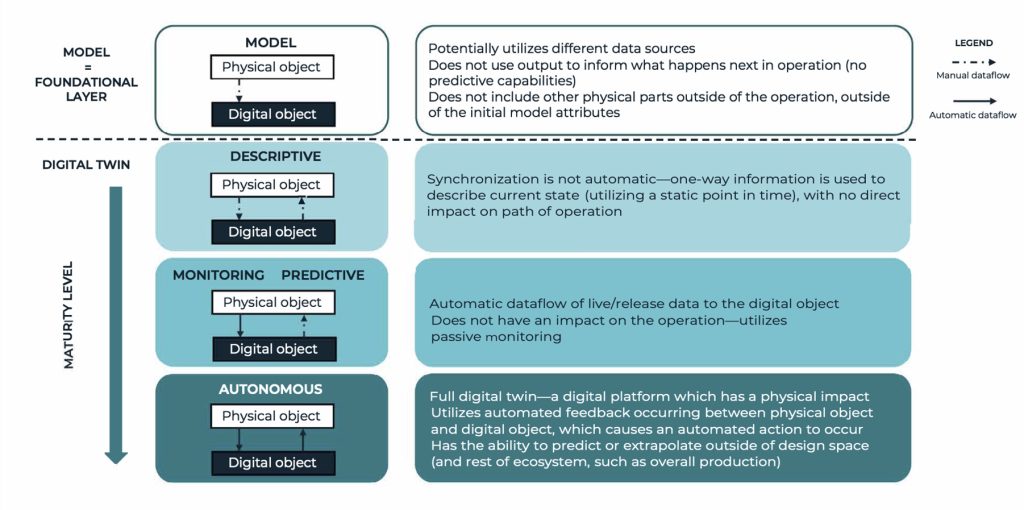
BioPhorum defines a digital twin as a digital replica of a physical asset, synchronised at a specified frequency and fidelity, enabling bidirectional connection to simulate, predict, and inform decisions based on real-world conditions. The novelty of this definition lies in its integration of a maturity model, acknowledging that digital adoption is a spectrum, not an absolute. Importantly, the model aligns with functional tracks, ensuring that the twin capabilities are embedded across the entire value chain.
This maturity model clarifies that basic tools, often confusingly termed ‘digital models’ or ‘digital shadows,’ are actually just nascent digital twins. The spectrum moves from a static, descriptive state, through a monitoring/predictive state, to the fully mature, autonomous state, where the twin delivers automated feedback that physically impacts the operation. By embracing this spectrum and viewing the definition as encompassing the entire drug product lifecycle, we gain the clarity needed to advance unified technological and regulatory strategies.
BioPhorum Digital Twin Definition
While defining the digital twin is a crucial first step, it doesn’t clear the roadblocks on the path. The biopharma sector, traditionally risk-averse and heavily regulated, faces a myriad of complex challenges spanning technology, regulation, and culture, all of which threaten to stall the widespread adoption of these tools. Overcoming these hurdles requires just as much collaborative standardisation as the definition itself.
On the technological front, the development of digital twins requires robust technological infrastructure and capabilities. Depending on the level of digital maturity, facilities may struggle to implement these areas, making the barrier to entry high. Technology must also be user-friendly for digital twins to be adopted widely in the pharma industry. Complex systems with steep learning curves can hinder adoption.
As with any new technology, regulatory validation and acceptance can be viewed as a major hurdle to development and implementation. Organisations are often not willing or able to accept the financial risk involved in running a dual process side by side to provide appropriate data. However, by doing so, and by increasing the availability of use cases of digital twin applications alongside recognised and defined terminology, digital twin technologies stand a greater chance of being accepted at higher maturity levels.
Finally, we must address organisational and cultural inertia. Biopharma is a change-resistant industry, where there is a perceived risk of fixing something that already works. Implementing digital twins requires a substantial initial investment in both expertise and technology, and proving a clear value and return on investment for this unproven technology can be difficult. Underlying much of this resistance is a lack of trust, trust rooted in the fear of additional validation workload, the reliance on advanced models, and the risk of making consequential errors based on ‘bad data’.
To move forward, companies must perform clear cost-benefit analyses, invest in comprehensive change management plans, and address the acute lack of available qualified personnel and expertise across the industry. The BioPhorum team have looked into the benefits of implementing digital twin frameworks and are working on an integrated digital twin for product, process and plant. Recent analysis demonstrates that they deliver substantial multi-dimensional benefits across the value chain. Companies adopting these solutions have reported up to 33 percent fewer engineering runs per site, a 33 percent reduction in batch failures, and 50 percent faster tech transfer and decision making. The impact is not merely theoretical, it could be as much as a 230 percent return on investment per annum.
Despite challenges, the potential for digital twins in biopharma is profoundly bright. We are standing at the precipice of a future where operations are not just monitored, but truly autonomous and optimised, leading to tangible improvements in cost, speed, and quality. The most significant catalyst in this evolution will be the deep integration of Artificial Intelligence (AI).
Advances in AI will revolutionise digital twin development, allowing for the creation of 3D models and dynamic simulations via simple prompts, reducing reliance on specialised technical personnel. AI will also generate accurate physics-based models, enhancing simulation precision. Regulatory acceptance of digital twin blueprints will standardise practices and foster trust. Enhanced data-sharing among all stakeholders, including regulators, will streamline processes and improve outcomes. User-friendly interfaces will ensure accessibility, enabling executive leadership to engage with digital twins and leverage insights for strategic decision-making. This evolution will make digital twins indispensable for innovation and efficiency in pharma.
But this autonomous future, with all its promise, remains fundamentally constrained by our present disunity. We cannot expect fragmented terminology to support a cohesive digital infrastructure. To ensure digital twins achieve their full maturation into high-value, autonomous assets, we must eliminate the lack of consensus on ‘digital twins’ that is hindering progress across pharma.
Download BioPhorum’s full paper, ‘Defining Digital Twins in a Biomanufacturing Environment’ here
* Andrea Johnston, PhD, is Senior Phorum Director at BioPhorum, accountable for outcomes and performance across a group of four Phorums and driving strategic growth, cross-Phorum collaboration, and industry thought leadership. She leads programmes that connect more than ninety initiatives and thousands of subject-matter experts to accelerate progress in biopharmaceutical manufacturing.
Previously Associate Director at CMAC, Andrea’s research career pioneered automated crystallisation and machine-learning applications, building on an AstraZeneca-sponsored PhD in advanced analytics. She joined BioPhorum in 2022 to lead Technology Strategy and has since scaled member value and impact across international collaborations.
** BioPhorum is a collaboration of industry leaders and subject matter experts working together to pool knowledge, best practice, and ideas – these outputs lead to results that effect real progress.
BioPhorum enables the global biopharmaceutical industry to connect, collaborate and accelerate progress for the benefit of all.


