Market Access & Health Technology Assesment: Saudi Arabia
Market Access & Health Technology Assesment: Saudi Arabia is a must-have asset for any company operating in Brazil or looking to enter the market.
Prepared in association with Youssry Saleh Law Firm, a leading Saudi Arabia law firm.
October 2025
1. Healthcare System and Funding: Saudi Arabia
1.Please make a general introduction to the public health sector in your country and its organization
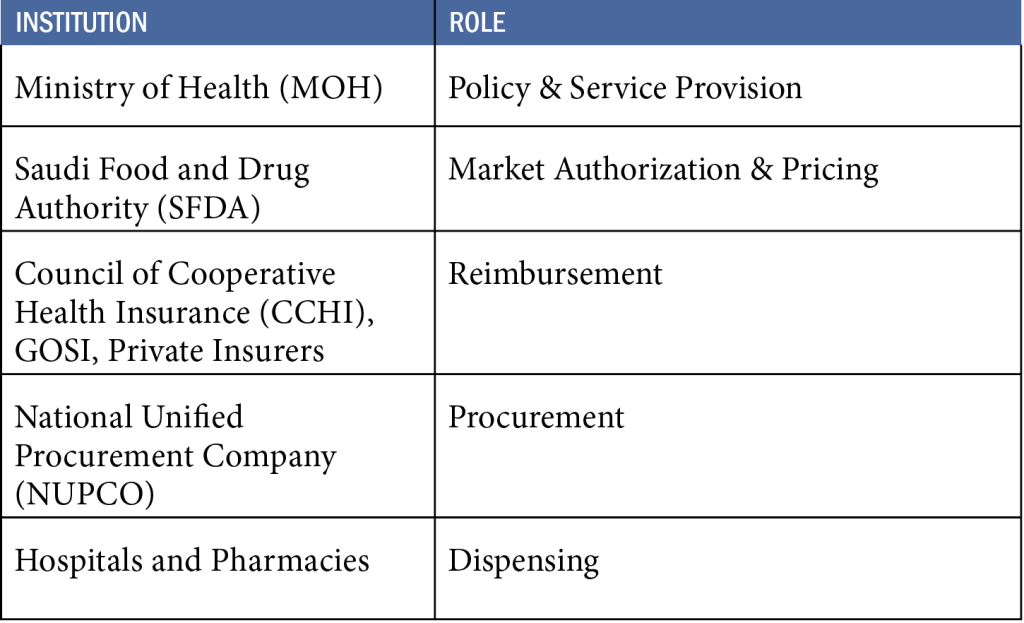
Saudi Arabia provides universal health coverage primarily funded by the government through a tax-based model. The healthcare system is organized in a tiered structure led by the Ministry of Health (MOH), which operates:
• Over 287 public hospitals
• More than 2,400 primary healthcare centers
The MOH is responsible for:
• Setting national health policies and regulations
• Licensing both public and private healthcare providers
• Delivering the majority of healthcare services
In parallel, other government entities such as the Ministry of Defence, National Guard Health Affairs, and Ministry of Interior operate separate healthcare networks for their employees.
Under Vision 2030, Saudi Arabia is transitioning toward corporatized Health Clusters, aiming to:
• Enhance efficiency
• Promote private sector participation
• Increase the share of private hospital beds from 23% (2023) to 68% by 2030
Healthcare Financing Sources:
1. State Budget: Approximately 60% of total health expenditure
2. Mandatory Cooperative Health Insurance (CCHI): Required for expatriates and Saudi nationals working in the private sector
3. Out-of-Pocket Payments: Gradually decreasing in line with Universal Health Coverage (UHC) goals
2. Please provide any infographics including
a. Stakeholders Involved
b. Required Data for Market Access
c. Market Access Process Timeline (Simplified)
1. Dossier Submission to SFDA
2. SFDA Technical Review (Typically 90–210 days)
3. Health Technology Assessment (HTA)
> Becomes mandatory starting July 2025
4. Pricing Committee Evaluation & Approval
5. Reimbursement Listing by CCHI or MOH (if applicable)
6. NUPCO Tender & Centralized Procurement
7. Product Distribution to Public Healthcare Facilities
Note: Infographic visuals such as stakeholder flowcharts, process arrows, and icons can be developed using official toolkits provided by MOH and SFDA for accurate representation.
Also from this Market Access & Health Technology Assessment
2. Healthcare Actors and Payers: Saudi Arabia
- Which are the administrations, bodies and institutions in charge of public health in your country and what are their respective responsibilities?
The Ministry of Health (MOH) is the primary body responsible for public health in Saudi Arabia. It sets national health policies, issues licenses to healthcare providers (public and private), oversees public hospitals and primary care centers, and regulates public health programs.
Its legal foundation lies in the Health Law (Royal Decree No. M/1, 2023) and the MOH Charter, which define its mandate as the lead coordinator and regulator of the national health system. Additional public health services are offered through parallel systems operated by the Ministry of Defence, National Guard Health Affairs, and Ministry of Interior for their respective populations.
2. Which are the administrations, bodies and institutions in charge of drug approvals in your country and what are their respective responsibilities?
The Saudi Food and Drug Authority (SFDA) is the sole competent authority for pharmaceutical registration. It evaluates the quality, safety, and efficacy of pharmaceuticals, vaccines, and biologics through a structured market authorization process. This includes technical reviews, Good Manufacturing Practice (GMP) inspections, and regulatory data assessment.
Its authority is defined by SFDA Law (2007). All medicinal products must be approved by SFDA before being marketed in the Kingdom.
3. Which are the administrations, bodies and institutions that qualify as “payers” in your country and what are their respective responsibilities?
Saudi Arabia’s healthcare system is supported by multiple payers:
- The Council of Cooperative Health Insurance (CCHI) oversees mandatory insurance schemes for private-sector workers and expatriates. It defines the Unified Benefits Package, supervises Third-Party Administrators (TPAs), and resolves reimbursement disputes in accordance with the Cooperative Health Insurance Law (Royal Decree M/10, updated 2023).
- The General Organization for Social Insurance (GOSI) purchases healthcare services for government-employed contributors under the Social Insurance Law (Royal Decree M/33, 2000).
- The MOH finances public healthcare services using state budget allocations.
- Over 27 private insurance companies, licensed by CCHI, are responsible for providing health coverage under regulated schemes.
4. Which are the administrations, bodies and institutions in charge of pricing decisions in your country and what are their respective responsibilities?
Drug pricing decisions fall under the jurisdiction of SFDA’s Pricing Committee, which applies a combination of:
- External Reference Pricing (ERP) using a basket of 34 countries
- Internal price comparison
- Value-based considerations
Final prices for ex-factory, wholesale, and pharmacy levels are determined based on SFDA’s 2023 Drug Pricing Guidelines.
5. Which are the administrations, bodies and institutions in charge of reimbursement decisions in your countries and what are their respective responsibilities?
Reimbursement responsibility is divided between:
- CCHI, which determines which medicines and services are covered under the insurance framework.
- MOH, which lists reimbursed drugs for public-sector use through internal formularies.
- Products must be both SFDA-priced and CCHI-listed to be reimbursed and procured by public buyers.
6. Which are the administrations, bodies and institutions in charge of Health Technology Assessment in your countries and what are their respective responsibilities?
As of July 2025, submission of economic evaluation dossiers becomes mandatory for high-cost or high-priority medicines seeking market access in Saudi Arabia.
This process is jointly governed by SFDA and CCHI, based on the HTA Guidelines (2024), and includes:
- Cost-effectiveness analysis
- Budget impact modeling
MOH also plays a coordinating role in integrating HTA into procurement and reimbursement pathways.
7. Which are the administrations, bodies and institutions in charge of public procurement and tendering in your country and what are their respective responsibilities
The National Unified Procurement Company (NUPCO) is the exclusive entity responsible for centralized public procurement of pharmaceuticals, vaccines, and medical devices.
NUPCO operates under the Council of Ministers Decision No. 7 (2009) and manages:
- National tenders
- Framework agreements
- Supply chain operations for MOH and other public entities
Only products approved by SFDA and listed by CCHI/MOH are eligible for tendering.
8. What are the other actors of significance with regards to market access in your country and what are their respective responsibilities?
Several other entities support or influence market access:
- The Ministry of Finance allocates national health budgets
- SASO (Saudi Standards, Metrology and Quality Organization) sets packaging and technical standards
- KACST (King Abdulaziz City for Science and Technology) supports R&D funding for health innovation
- The Vision 2030 Delivery Unit monitors targets for private sector participation in healthcare transformation
Also from this Market Access & Health Technology Assessment
3. Post Market-Approval Processes and Regulations: Saudi Arabia
- What are the pricing models, processes and principles for originator drugs?
Originator drugs (innovator medicines) are priced by the Saudi Food and Drug Authority (SFDA) following the principles outlined in its 2023 Drug Pricing Guidelines. The pricing process includes:
- External Reference Pricing (ERP): SFDA compares prices with a reference basket of 34 countries.
- Internal Reference Pricing (IRP): Pricing is compared to existing alternatives in the local market.
- Cost-based evaluation: Includes manufacturing costs, value-added services, and public health need.
- Therapeutic value assessment: High clinical benefit may justify a premium price.
The company submits a complete pricing dossier, and the final decision is issued by SFDA’s Pricing Committee. No product can be reimbursed or procured until a formal SFDA price is approved.
2 . What are the pricing models, processes and principles for generics and biosimilars drugs?
Generic and biosimilar products are subject to differentiated pricing rules to encourage market competition and cost savings. SFDA applies the following principles:
Price Cap Model:
Generics are priced at a fixed percentage below the originator (e.g., 70%–80% of reference price).
Biosimilars may follow a similar model but with flexibility depending on manufacturing complexity.
Stepwise Reduction:
If multiple generics exist, prices are reduced further with each new entry.
No Premium Pricing:
Generics and biosimilars are generally not allowed price premiums unless exceptional clinical or supply-chain value is demonstrated.
Streamlined Process:
SFDA may offer an expedited pricing review for bioequivalent generics.
Once priced, generics and biosimilars are eligible for listing and reimbursement under the same formulary systems as originators.
3. What are the reimbursement approval processes and principles for originator drugs?
Once priced by SFDA, originator drugs can be submitted for inclusion in the Unified Drug List (UDL) and Unified Drug Formulary (UDF), which are managed by CCHI and MOH.
The process includes:
- Review of clinical effectiveness, safety, cost-effectiveness, and budget impact.
- As of July 2025, submission of Health Technology Assessment (HTA) will be mandatory for high-cost or high-priority drugs.
- If approved, the drug is listed with an assigned co-payment level (typically 0–20 SAR).
- Once listed, it becomes eligible for public procurement through NUPCO tenders.
Only drugs that are both SFDA-priced and CCHI-listed are eligible for reimbursement.
4. What are the reimbursement approval processes and principles for generics and biosimilar drugs?
Generics and biosimilars follow a similar reimbursement pathway to originator drugs, but with specific considerations:
Cost advantage is key:
Reimbursement is often prioritized if the product offers significant savings compared to originators.
Therapeutic equivalence:
SFDA requires proof of bioequivalence (for generics) or comparability (for biosimilars) before they are considered for reimbursement.
Fast-track listing:
CCHI may apply simplified listing for certain generics already included in UDL/UDF.
Co-payment policies:
Generic drugs often have lower patient co-payment rates to encourage usage.
Formulary tiering:
Generics and biosimilars may be placed in preferred tiers unless the originator has exclusivity or documented superior clinical value.
Like originators, they must be SFDA-approved, priced, and listed by CCHI to be eligible for public procurement and insurance coverage.
Also from this Market Access & Health Technology Assessment
4. HTA Dossiers: Saudi Arabia
- Have local authorities published recommendations surrounding value assessment dossiers? (If yes please add link)
Yes. The SFDA published the Economic Evaluation Studies Guideline (PDF, July 10, 2024), which outlines comprehensive methods and includes a checklist. Followed by the Pharmacoeconomic Submission Portal Manual (January 2025), detailing dossier preparation and submission protocols.
- Have local Authorities published guidelines surrounding value assessment dossiers? (If yes please add link)
Yes, included in the same SFDA documents:
- Economic Evaluation Studies Guideline (July 2024)
- EES Submission Checklist
- Portal Manual (2025)
These provide structured guidance for dossier development and submission.
- Have local authorities published official guidelines surrounding the submission of value assessment dossiers? (If yes please add a link)
Yes. The Portal Manual (2025) is officially issued by SFDA and covers the EES portal use, data formats, timelines, and administrative requirements.
- Describe the overall process of preparing and submitting a HTA dossier in your country.
The process is as follows:
- Optional pre-submission meeting with SFDA’s HTA unit to align on comparator, model structure, and data sources.
- Dossier submission via SFDA’s e-services in eCTD format, including economic model plus appendices.
- Administrative validation (about 10 days) for completeness.
- Technical review rounds (typically 60 days with clock-stops for clarifications).
- HTA committee assessment, price negotiation, and issuance of an Outcome Letter.
- Outcomes are forwarded to MOH/CCHI for further reimbursement listing.
- Describe the overall content of the HTA dossier in your country.
The dossier must include:
- Executive summary and objectives
- Disease and treatment landscape
- Justification and choice of comparator
- Clinical efficacy and safety data
- Economic evaluation module (CEA/CUA/CMA)
- Budget Impact Analysis (covering 5+ years)
- Sensitivity analyses (deterministic and probabilistic)
- Summaries of international HTA reports
- Local real-world evidence plan
- Appendices: price certificates, model files, data sources
- Which are the questions to focus on when preparing a HTA dossier in your country?
Your dossier should explicitly answer:
- Does the product offer a clinically significant added benefit compared to SOC?
- Is the incremental cost-effectiveness ratio (ICER) below the reference threshold of 150,000 SAR per QALY?
- What is the projected 5-year budget impact for MOH and insurers?
- Are the model inputs localized for cost, epidemiology, and resource use?
- Which are the other strategic considerations to take into account when preparing a HTA dossier in your country?
Strategic factors to include:
- Engage early with SFDA to agree on comparator and model approach
- Align dossier timing with NUPCO tender cycles
- Highlight alignment with Vision 2030 objectives (e.g., tech transfer, local manufacturing)
- Propose confidential discount and risk-sharing scenarios to meet willingness-to-pay (WTP) thresholds
- Prepare a post-launch real-world evidence plan to support future HTA reassessments or re-pricing
Also from this Market Access & Health Technology Assessment
5. Data Requirements: Saudi Arabia
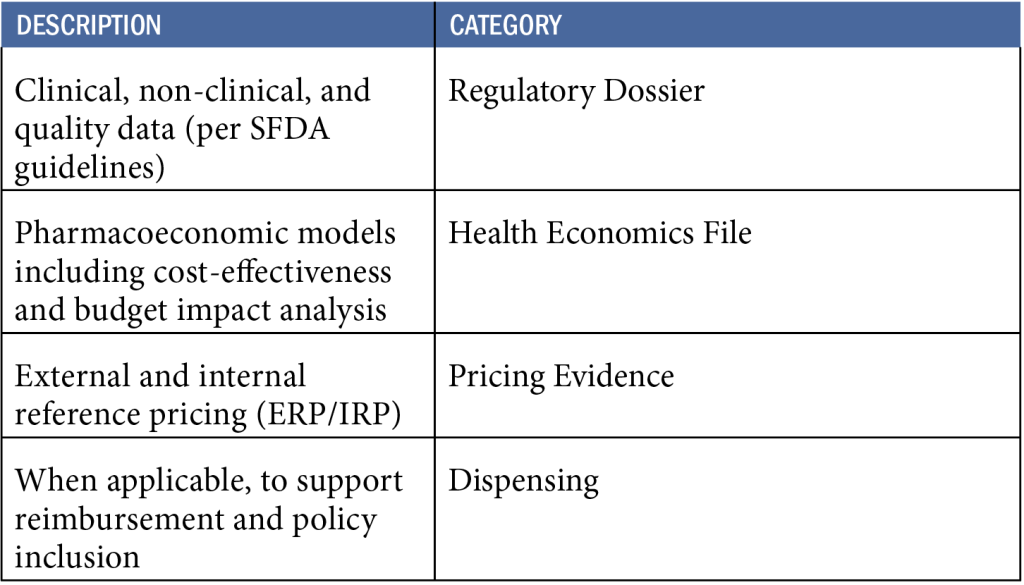
- In addition to the clinical data obtained through clinical studies please list the data required for
a. Market approval
In addition to clinical trial data, the following documents are required for market authorization in Saudi Arabia:
- Quality/CMC Module: Includes full GMP certificates (PIC/S or WHO type) and a local QP release plan.
- Non-clinical studies: Including toxicology and pharmacology data.
- Risk Management Plan (RMP) and a pharmacovigilance (PV) system master file validated for KSA.
- Arabic labelling: Mock-ups of patient leaflets and packaging with local language.
- Stability data: Minimum 6 months of data for IV products.
- Certificate of Pharmaceutical Product (CPP) issued by a reference country.
- Evidence of compliance with Saudi-specific excipient limits (e.g. ethanol, sugar).
All documents must be submitted via the SFDA SDR Portal using the eCTD format.
b. Pricing Decisions
In addition to clinical data, pricing decisions require:
- Notarized ex-factory price certificates from all 16 countries in the ERP basket.
- Commercial invoice for the first production batch.
- ERP pricing template and a full EES dossier (PDF).
c. Reimbursement Decisions
To support reimbursement approval, the following additional data must be submitted:
- Final HTA report from SFDA and the ICER.
- Five-year Budget Impact Model, with cost comparison between public and private insurers.
- Comparative treatment analysis and an unmet medical need statement.
- Real-world evidence (RWE) and/or a risk-sharing proposal if uncertainties exist.
- Dispensing data at the pharmacy level, especially if the product was already sold in the private market.
Also from this Market Access & Health Technology Assessment
6. Managed Entry Agreements: Saudi Arabia
- Are there any Managed entry agreements in place in your country? (If so, please list them)
Yes, Saudi Arabia has implemented two types of Managed Entry Agreements (MEAs) under the Ministry of Health’s “Managed-Entry Agreement Policy” issued in January 2021. These include:
A. Finance-based Price/Volume Agreement
1. Fundamentals & rationale
- The Marketing Authorization Holder (MAH) agrees to apply tiered discounts once cumulative public volume exceeds specific thresholds.
- The Saudi Food and Drug Authority (SFDA) fixes the public list price, while the confidential net price is captured in the NUPCO contract.
- NUPCO monitors purchase volumes quarterly.
2. When to consider this MEA
- This agreement is used for high-budget, innovative drugs with predictable demand, such as diabetes drugs (e.g., GLP-1s).
3. Requirements
- Volume forecast accuracy within ±20%.
- Supplier must provide a performance bond worth 10% of the contract value.
- Electronic sales data must be submitted to NUPCO.
4. Impact on product uptake
- Facilitates faster listing in the national formulary at the public price while maintaining control over spending.
5. Challenges
- Under-forecasting may lead to rebates.
B. Outcome-based Risk-Sharing Agreement (Payment-by-Results)
1. Fundamentals & rationale
- Reimbursement is conditional upon achieving a predefined clinical outcome (e.g., ≥95% Sustained Virologic Response for hepatitis C treatments).
- Hospitals report patient-level outcomes to the MOH registry.
- Manufacturers must refund costs for non-responding patients.
2. When to consider this MEA
- Best suited for drugs with high clinical or economic uncertainty, such as first-in-class oncology therapies or Advanced Therapy Medicinal Products (ATMPs).
3. Requirements
- Existence of a robust real-world registry.
- Clear metrics and defined timelines for outcome assessment.
- Use of escrow or credit-note mechanisms for reimbursement.
4. Impact on product uptake
- Enables early access even in the presence of limited clinical data.
- Positive real-world outcomes accelerate adoption.
5. Challenges
- High burden of data capture.
- Potential disputes over outcome adjudication.
- Confidentiality of net pricing must be maintained.
Additional note:
Publicly disclosed examples include:
- Finance-based MEAs for insulin analogues and antiviral drugs.
- Outcome-based MEA piloted in 2024 for CAR-T therapy at King Faisal Specialist Hospital.
Also from this Market Access & Health Technology Assessment
7. Public Procurement and Tendering: Saudi Arabia
- Which are the main actors involved in public procurement and tendering?
In Saudi Arabia, the main actors in the public procurement and tendering process for pharmaceuticals include:
- NUPCO (National Unified Procurement Company): Acts as the central purchasing agent for the Ministry of Health (MOH), Ministry of Defense, National Guard, and universities.
- SFDA (Saudi Food and Drug Authority): Issues import permits and ensures quality control before products are released.
- Etimad e-Procurement Platform: Managed by the Ministry of Finance, used to publish tender announcements and contracts.
- Hospital Pharmacy and Therapeutics Committees: Involved in demand forecasting and delivery verification at the institutional level.
- What are the main characteristics of the public procurement and tendering system?
- Centralized & Electronic: All pharmaceutical tenders above SAR 1 million are processed through NUPCO’s e-tender portal.
- System Integration: NUPCO’s portal is synchronized with Etimad, the national procurement system.
- Supplier Requirements: Vendors must be accredited by NUPCO and hold valid SFDA marketing authorization and GMP certification.
- Evaluation Criteria: 60% based on price,
- 25% on quality and technical compliance,
- 15% on localization score (aligned with Vision 2030’s “Made in Saudi” initiative).
- Contract Structure: Multi-year framework agreements (2–3 years), including commitment to supply volumes and maintain strategic stock.
- Transparency: Awarded prices (excluding confidential MEAs) are made public on the NUPCO dashboard.
- Payment Terms: Suppliers are paid within 30 days post-delivery confirmation via the SADAD system.
- Appeals: Vendors can contest decisions via the Government Tenders & Procurement Law (2021) dispute committee.
Also from this Market Access & Health Technology Assessment
8. Expenditure Control and Cost-Containment Policies: Saudi Arabia
- Please describe the main cost containment policies in place in your country and their fundamental principles
a. Pricing and impact of generic/biosimilar approval
The SFDA applies automatic price reductions for generics and biosimilars:
- The 1st generic is priced ≥35% lower than the originator.
- The 2nd generic is ≥55% lower.
- Additional generics must be ≥10% cheaper than the previous one.
- Biosimilars are priced ≥30% lower than the reference biologic.
- When a generic is listed, the originator’s list price is reduced by 20%.
Legal reference: SFDA Pharmaceutical Pricing Rules (Oct 2023) – sfda.gov.sa
b. Clawback/Payback/Discounts/Rebates
According to the MOH MEA Policy (2021), manufacturers may be subject to:
- Invoice-level discounts.
- Tiered paybacks if total spend exceeds a set cap.
- Free-dose schemes.
Legal reference: MOH MEA Policy 2021 – moh.gov.sa
c. Price/Volume agreements in public tendering
NUPCO contracts include sliding-scale discounts based on delivered volumes.
- Breaching volume thresholds triggers automatic price reductions or rebates.
Legal reference: NUPCO Tender Manual – nupco.com
d. Price freezes and cuts
SFDA may freeze prices for up to 3 years after initial setting.
- Re-evaluation may lead to up to 30% additional price reductions.
Legal reference: SFDA Pricing Rules Article 27; Re-pricing Guidance 2024 – pharmaknowl.com
e. Post-launch monitoring of prescriptions/sales
- SFDA’s e-pharmacovigilance system and “Wasfaty” e-prescription platform monitor drug utilization and adverse events (ADRs).
- CCHI conducts audits and flags abnormal volume claims for price review.
Legal reference: SFDA Pharmacovigilance Law 2015 – PMC link
f. Generic substitution policies
Mandatory in public hospitals and most community pharmacies.
- Exception: Prescriber marks “no substitution.”
Legal reference: MOH Circular 2022; CCHI Formulary Rules 2024
g. At prescriber level (Incentives)
As part of Vision 2030 hospital KPIs:
- Prescribers with ≥80% generic prescribing receive CME credits.
- Outliers may be flagged for audits.
Legal reference: MOH Circular 2023
h. At retail level (Incentives)
- Pharmacies must stock the lowest-priced generics.
- SFDA mobile app allows patients to compare official ceiling prices.
Legal reference: SFDA Price-Display Regulation 2022
- Are there any other policies in place aiming at cost control via incentive programs targeting the different actors (pharma companies, wholesalers, retailers, prescribers etc)?
Saudi Arabia also encourages cost control via broader incentive schemes:
- “Made in Saudi” localization score (15% weight in NUPCO tenders) favors companies that locally manufacture or fill-finish drugs.
- Zero-tariff import duty (2022) on WHO-listed generics further reduces costs.
Also from this Market Access & Health Technology Assessment
9. HTA Decision Analysis Framework: Saudi Arabia
- Which are the health technology assessment (HTA) evaluation bodies and their responsibilities in your country?
The primary HTA authority is the Saudi Food and Drug Authority (SFDA), which hosts a dedicated HTA/Economic Evaluation Unit established in 2023.
Its main responsibility is to evaluate clinical and economic evidence (EES) for:
- New chemical entities
- Biologics
- Re-pricing of existing products
The Ministry of Health (MOH) uses HTA outcomes for hospital listing and budget allocation decisions.
The Council of Cooperative Health Insurance (CCHI) relies on SFDA’s HTA reports when assessing benefit package inclusion within the private insurance system.
- Do regulators require HTA studies in your country?
Yes. According to SFDA Economic Evaluation Guidelines (2024), submitting an HTA (EES) study is mandatory starting July 2025 for:
- New drugs and biologics
- Orphan drugs
- High-cost or re-priced drugs
- First-in-class or biosimilars targeting high-impact diseases
- Do payers require HTA studies in your country?
Yes. Both MOH and CCHI require HTA-based evidence to approve a product for inclusion in the Unified Drug List (UDL) or Formulary.
Without a valid HTA report, the product may be excluded unless a justified exception is granted.
- How are HTA assessments translated into pricing conditions in your country?
HTA outcomes directly influence SFDA’s Pricing Committee decisions.
They consider:
- Incremental cost-effectiveness ratios (ICERs)
- Budget impact
- Whether the proposed price is aligned with the clinical value
- Confidential discounts, risk-sharing or clawback agreements may be applied
A standard cost-effectiveness threshold of 100,000–150,000 SAR per QALY is typically referenced.
- How are HTA assessments translated into reimbursement conditions in your country?
Positive HTA outcomes lead to:
- Product listing in UDL/UDF
- Assignment to the lowest co-payment tier (usually 0–20 SAR)
- Eligibility for NUPCO tenders
Unclear or negative HTA results may lead to access restrictions, conditional reimbursement, or product rejection.
- Which are the evaluation criteria, processes or models and analyses framework used for HTA in your country?
Core evaluation criteria include:
- Clinical effectiveness
- ICER
- Budget impact
- Disease severity
- Manufacturing cost
- Unmet medical need
Preferred analytical models include:
- Cost-utility analysis (CUA)
- Cost-effectiveness analysis (CEA)
- Cost-minimization (CMA) and cost-consequence models
- Decision-analytic frameworks such as Markov models for chronic/lifetime conditions
A minimum 3% discount rate is applied for both cost and outcome projections.
- What is the methodology used in your country for HTA assessment?
SFDA’s EES Guidelines require:
- MOH or payer perspective
- Use of local comparator drugs
- Local epidemiological and price data
- Deterministic and probabilistic sensitivity analyses
- Inclusion of a Budget Impact Analysis (BIA) covering a minimum of 3–5 years
- Which are the other decisions impacted by the assessed outcome in your country?
HTA outcomes also impact:
- NUPCO tender eligibility and pricing caps
- Product’s “essential/restricted” status in MOH lists
- Re-pricing triggers for renewal
- Volume-based or risk-sharing contract eligibility
- Does your HTA review or inquire other international HTAs during the assessment process? If so, which ones are the usual partners?
Yes. SFDA requires a summary of at least two international HTA outcomes for benchmarking.
Also from this Market Access & Health Technology Assessment
10. Price Control and Reference Pricing Systems: Saudi Arabia
1.Price Control
- How does price control at ex-factory prices work in your country?
Ex-factory prices are strictly regulated by the Saudi Food and Drug Authority (SFDA) using External Reference Pricing (ERP). SFDA sets a Ceiling CIP price based on the lowest value from a basket of 34 reference countries. Manufacturers cannot exceed this ceiling when submitting pricing proposals.
Legal basis: SFDA Drug Pricing Rules (2023
2. How does price control at the wholesale level work in your country?
Wholesale prices are capped based on a margin system:
- 15% margin on the ex-factory price up to 100 SAR
- 10% for the portion between 100–400 SAR
- 5% for the portion beyond 400 SAR
Legal basis: SFDA Pricing Rules – Annex 4
3. How does price control at the retail pharmacy level work in your country?
Retail pharmacy prices follow the Maximum Retail Price (MRP) model:
- MRP must be printed on the outer pack
- Pharmacists apply a standard margin of 20–25%
- SFDA also regulates discounts on bundled items
- Pharmacies are required to display official SFDA price lists
Legal basis: MOH/SFDA Unified Circular 2022
2.External Reference Pricing (ERP)
- Is there a system of external reference pricing (ERP) in place in your country?
Yes – ERP is mandatory for all pharmaceuticals including biologics and high-cost therapies.
- When and/or how often is ERP activated?
ERP is triggered:
- Every 3 years
- Or earlier, if prices in 5+ reference countries drop more than 20%
- What is the legal framework of the ERP in place in your country?
ERP is governed by the SFDA Pharmaceutical Pricing Rules, latest version consolidated in Q4 2023.
- What is the composition of the country basket?
The ERP basket includes 34 countries, including:
- Australia, UK, USA, Canada, France, Germany, Netherlands, Hungary, South Korea, Spain, Portugal, Switzerland, and others
- Describe the price calculation and selection for reference products.
SFDA selects the lowest publicly available factory price (CIP) from the basket, or calculates an average if data is missing. Preference is given to official government sources or verified pricing databases.
- How often does the price need to be updated?
Prices must be re-evaluated:
- Every three years, or
- Earlier if an ERP trigger is met (≥5 countries drop price by 20%)
- How do the “price List”/catalogues from references countries work in your country?
SFDA requires manufacturers to submit validated price certificates. It cross-checks foreign prices with sources like:
- Australian PBS, French CEPS, German Lauer-Taxe, etc.
Verified prices are published on SFDA’s public database.
3.Internal Reference Pricing (IRP
- Is there an internal reference pricing (IRP) system in your country?
Yes – IRP applies to drugs with therapeutically equivalent alternatives or similar clinical use.
- What is the legal framework of the IRP in place in your country?
Governed under Article 6 of SFDA Pricing Rules and the “Similarity Group” policy (2021).
- When and/or how often is IRP activated?
IRP is triggered once a generic or biosimilar enters the market.
SFDA then adjusts the originator’s price downwards to align with the lowest-priced equivalent, and re-evaluates prices annually.




